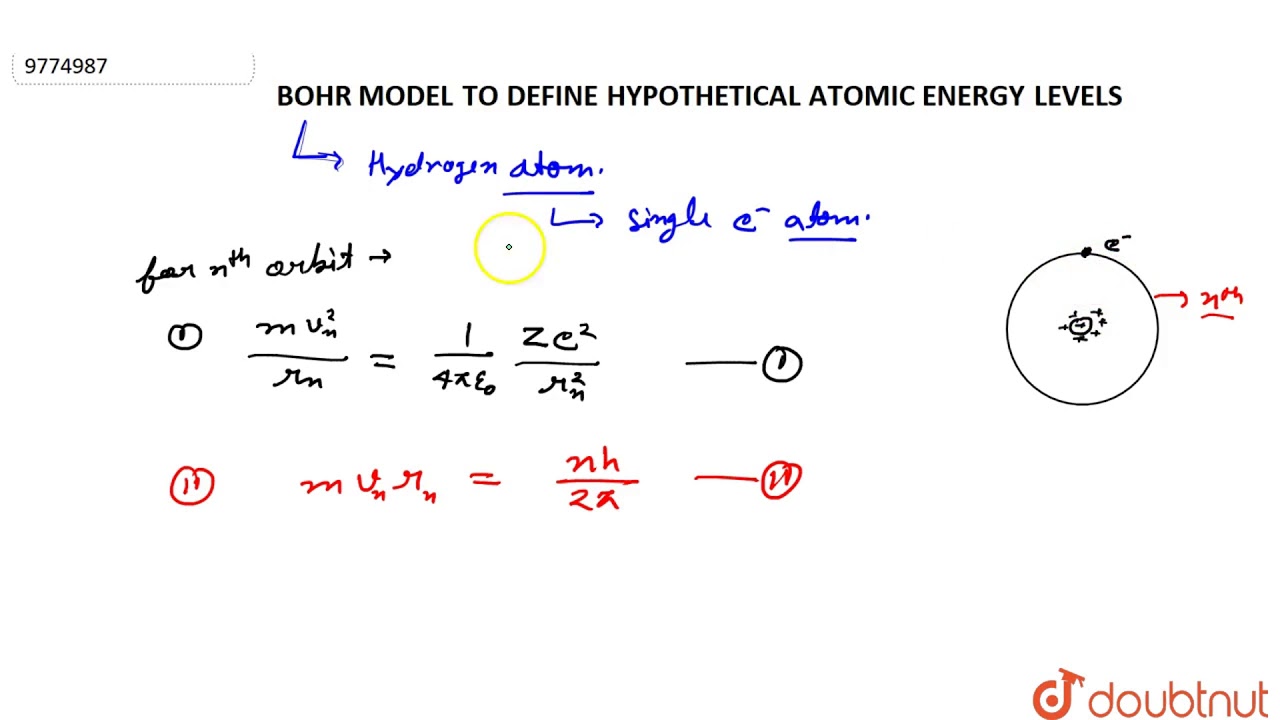
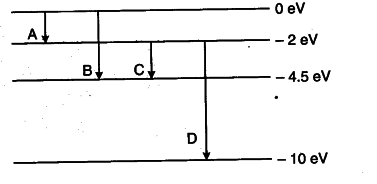
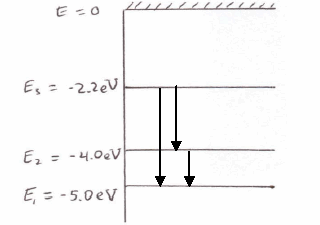
The energy levels of a hypothetical atom are shown below which of the shown transition will result in the emission of a photon of wavelength 275nm which of these transitions correspond to
a) The energy levels of a hypothetical hydrogen-like atom are shown in the figure. Find out the transition, - Sarthaks eConnect | Largest Online Education Community
An energy-level diagram for a hypothetical atom is shown above. - Sarthaks eConnect | Largest Online Education Community

Schematic energy levels of a hypothetical atom with configurations d 2... | Download Scientific Diagram

OneClass: Be sure to answer all parts. Consider the following energy levels of a hypothetical atom: E...