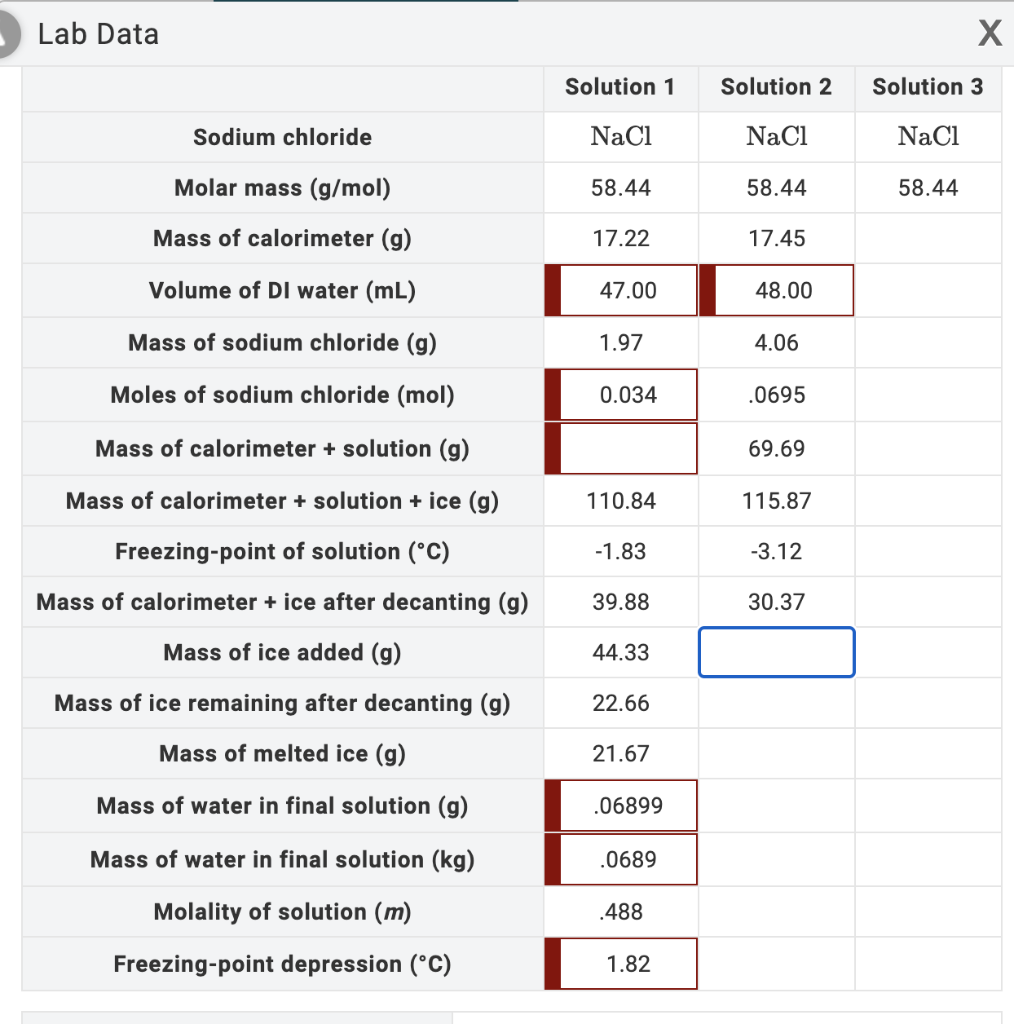
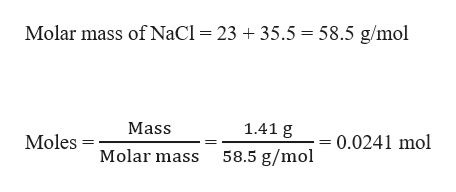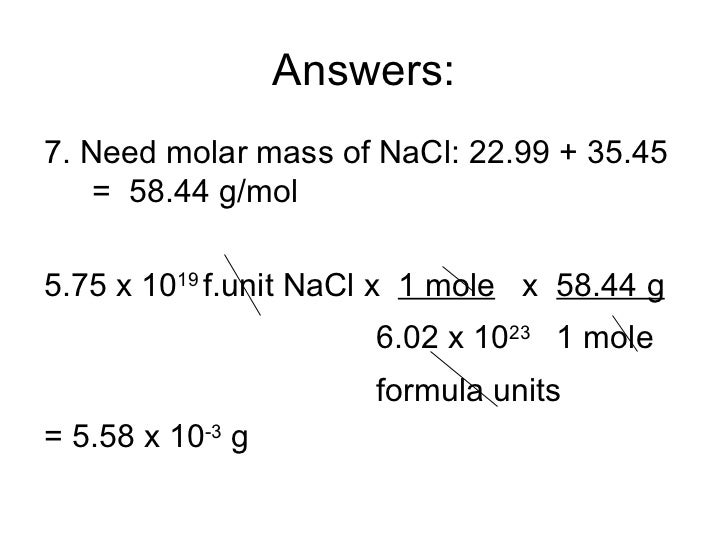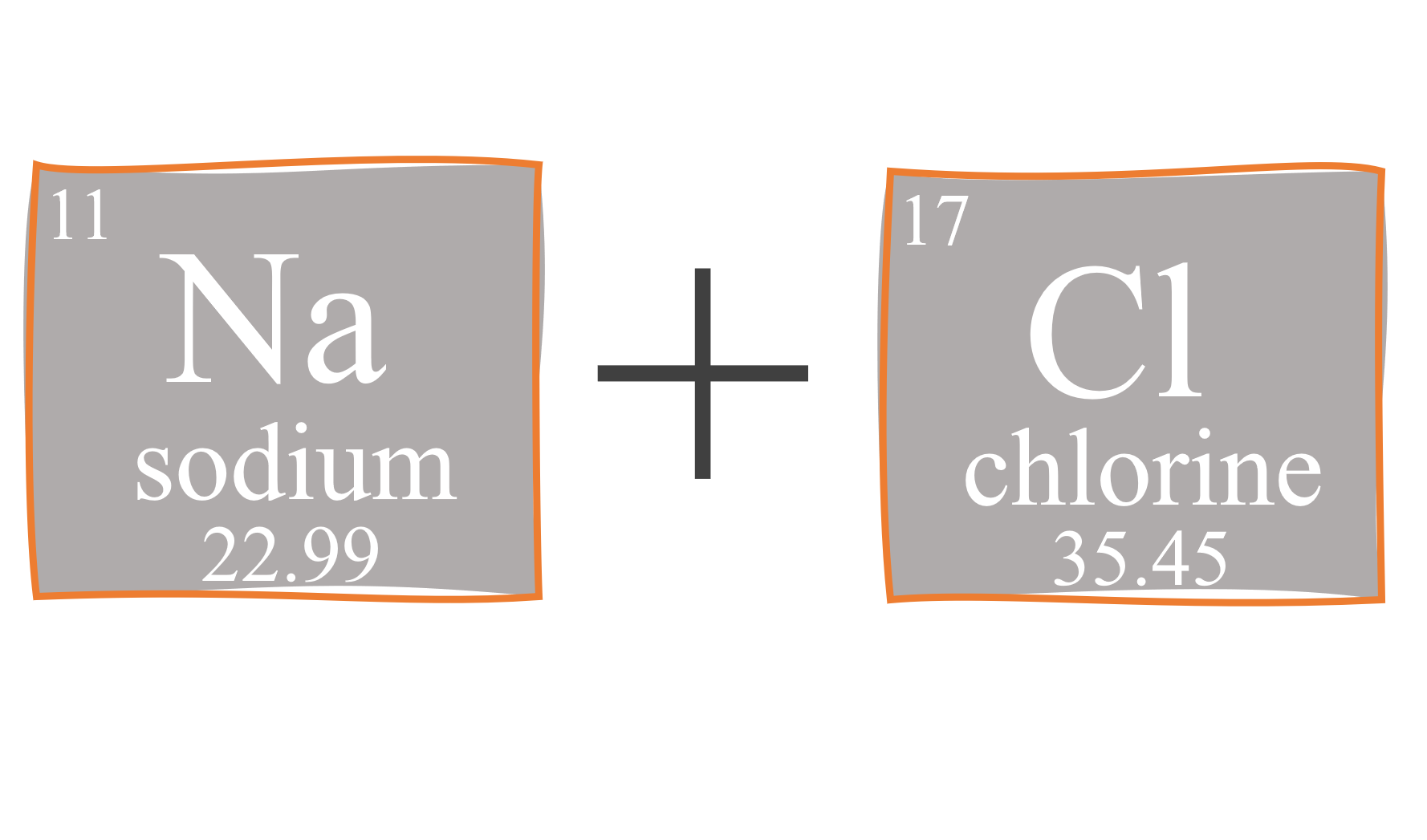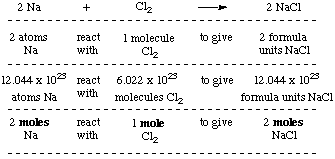25. The molecular weight of NaCl determined by studying freezing point depression of its 0.5% aqueous solution is 30. The apparent degree of dissociation of NaCl is (1) 0.95 (2) 0.45 (3) 0.60 (4) 0.35

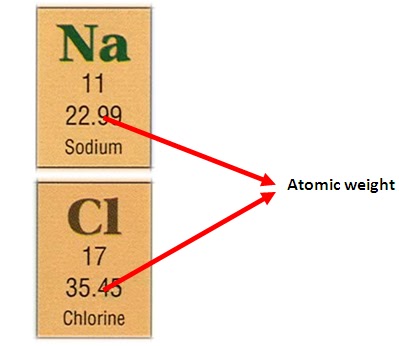
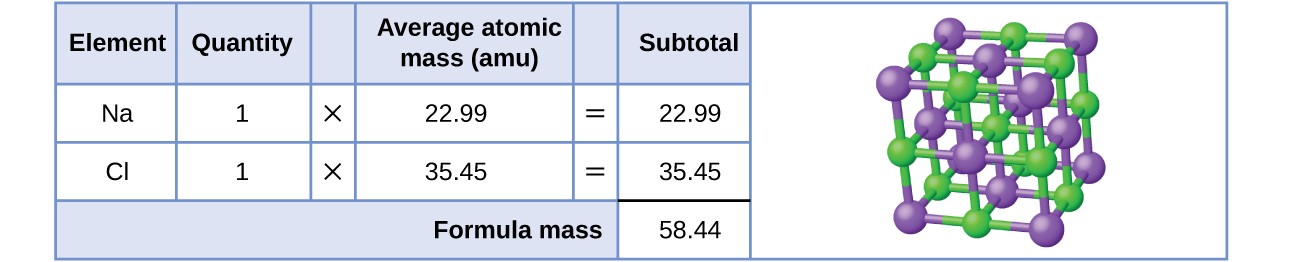
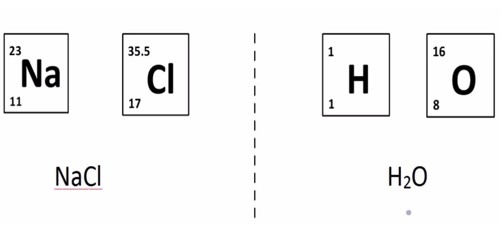
Chemistry 3/11/15 Formula Mass and Molar Mass. Drill Make sure you have a calculator!! What is the atomic mass of sodium? What about sodium chloride? - ppt download
Formula mass of NaCl is 58.45 g mol^-1 and density of its pure form is 2.167 g cm^-3. The average distance - Sarthaks eConnect | Largest Online Education Community

Dependence on the NaCl concentration of the apparent molar mass (a) and... | Download Scientific Diagram

The molar mass of table salt (NaCI) is 58.5 g/mol. What mass of salt is equivalent to 3.00 moles - Brainly.com

Densities of phonon states of a crystal of the NaCl structure for the... | Download Scientific Diagram

The reaction, 2A(g) + B(g) 3C(g) + D(g) , is begun with concentration of A and B both at initial value of 1 M . When equilibrium is reached, the concentration of